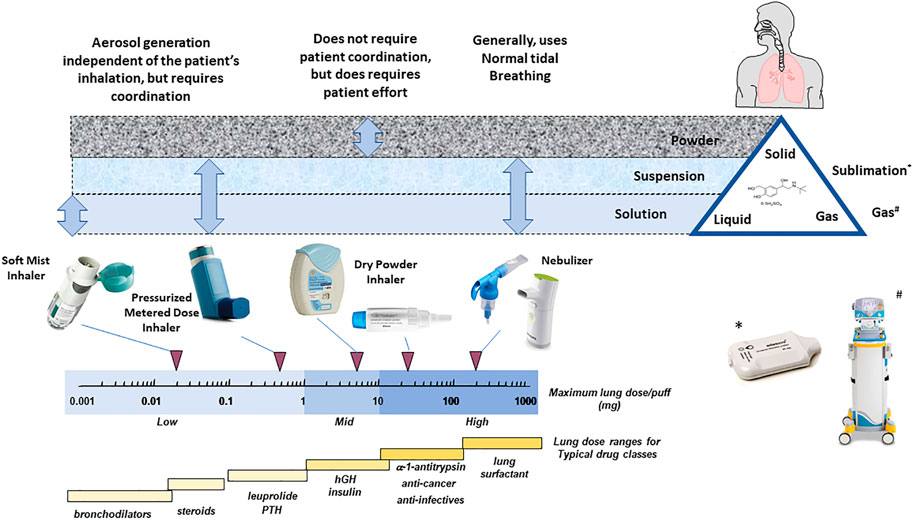
Frontiers | Half a Century of Technological Advances in Pulmonary Drug Delivery: A Personal Perspective

Pharmaceuticals | Free Full-Text | Effect of MDI Actuation Timing on Inhalation Dosimetry in a Human Respiratory Tract Model

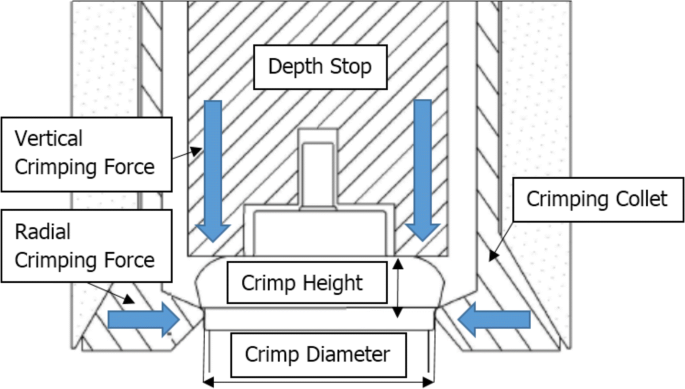
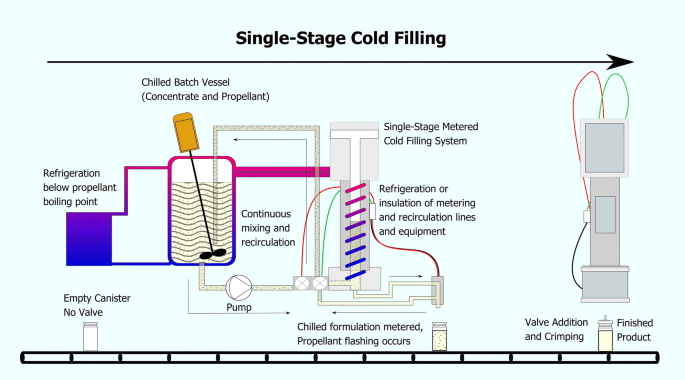
PROCESS VALIDATION AND REGULATORY REQUIREMENTS OF METERED-DOSE INHALERS: AN OVERVIEW | Semantic Scholar
![PDF] Formulation and product development of pressurised metered dose inhaler: An overview | Semantic Scholar PDF] Formulation and product development of pressurised metered dose inhaler: An overview | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c4f3be50a4301bab07e0101b7840fc36a7348944/4-Table2-1.png)
PDF] Formulation and product development of pressurised metered dose inhaler: An overview | Semantic Scholar
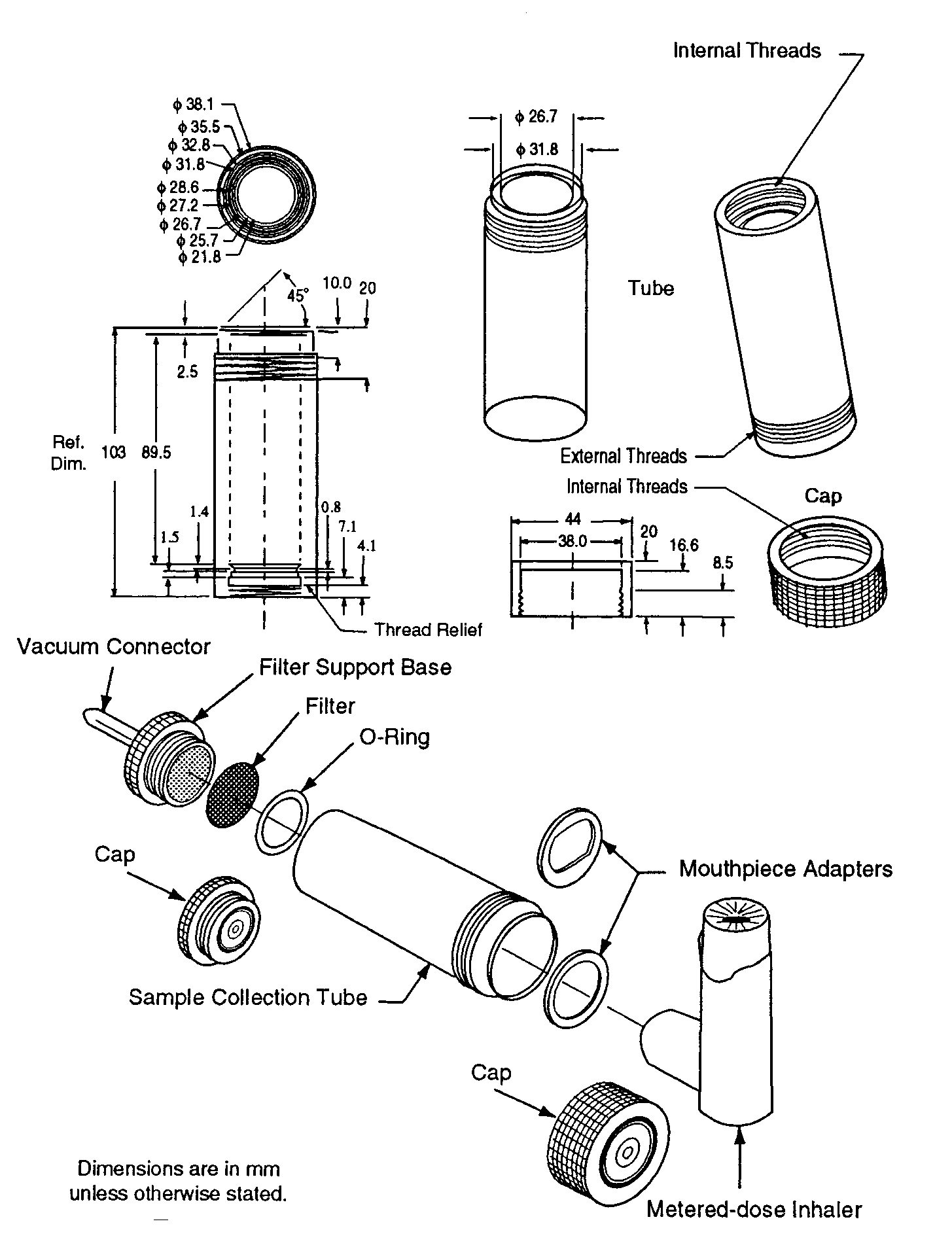
METERED-DOSE INHALERS AND DRY POWDER INHALERS, chemical structure, molecular formula, Reference Standards

Scientific and regulatory activities initiated by the U.S. food and drug administration to foster approvals of generic dry powder inhalers: Quality perspective - ScienceDirect
![PDF] Formulation and product development of pressurised metered dose inhaler: An overview | Semantic Scholar PDF] Formulation and product development of pressurised metered dose inhaler: An overview | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c4f3be50a4301bab07e0101b7840fc36a7348944/2-Figure1-1.png)
PDF] Formulation and product development of pressurised metered dose inhaler: An overview | Semantic Scholar
Pandita Rachna et al. IRJP 2011, 2 (12), 114-116 INTERNATIONAL RESEARCH JOURNAL OF PHARMACY ISSN 2230 – 8407 Review Art